
Fulton MSC Force Surgeon. Since its inception, the Anthrax Vaccination Immunization Program for the military services has gone through numerous phases. But the purpose of the program has never wavered from. DoD News Briefing Monday, December 13. Also Participating: Dr. Secretary Cohen and Chairman Shelton both completed the vaccination program. ASA Newsletter - Anthrax Vaccine Immunization Program. This is the second in a series of article exploring the. The first article appeared in ASA 9. A similar article by Dr. Nass has appeared in Infectious. Disease Clinics of North America, volume 1. Number 1, March. 1. Biological Warfare and Vaccines: Anthrax by Meryl Nass, M. D. A Brief History of Anthrax Vaccine Development. Pasteur, Toussaint and Greenfield developed the first animal. There are rare animal outbreaks in the. US, and less than one human case per year (9. 
The Sterne vaccine strain lacks the plasmid p. X0. 2, which codes. This fragment remains on the cell surface. EF or LF. A heptamer of. PA- LF or PA- EF complex creates a pore in the cell membrane. Collier. RJ and Liddington R. Model presented at the 3d International. Conference on Anthrax, Plymouth UK, 9/9. As noted in ASA 9. PA is critical because. PA with either LF or EF that is necessary. Therefore, if an immune response to PA can. PA. However, such broad immunity is not necessarily induced. Current Human Vaccines. Human vaccines were developed in the Soviet Union by 1. US and. Great Britain in the 1. The current. US vaccine was formulated in the 1. Russia and China use live attenuated strains for their human. The Chinese and Russian vaccines may be given by aerosol. The Russian vaccine was manufactured at. George Eliava Institute of Bacteriophage, Microbiology and. Virology in Tblisi, Georgia until 1. Nina Chanishvili, Ph. D. personal communication, June 1. The efficacy of the live. Russian vaccine is reported to be greater than that of the killed. US or British vaccines. As with the Sterne vaccine strain, each lacks. X0. 2. These strains are composed chiefly. PA (5. 2) with small amounts of EF and LF. Whether or not the EF and LF contribute. The British vaccine consists. EF and LF than the US vaccine (8. The US. vaccine uses aluminum hydroxide (alhydrogel) to adsorb PA, and. 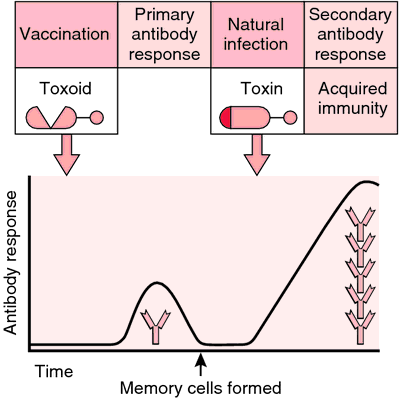
The mechanism. of action of the adjuvant is not entirely understood. According. to Hambleton and Turnbull, . 
Anthrax Vaccine Immunization Program (AVIP) Prepared by Anthrax Vaccine Immunization Program (AVIP) Agency. Six independent civilian reviews since 1978 unanimously affirm the value of anthrax vaccination. The Immunization Program Leaders Course. Influenza Infection and Influenza Vaccines 01 Aug 16; Anthrax Infections and Anthrax Vaccine 25 Jul 16. 2016–2017 Seasonal Influenza Vaccination Program 11 Aug 16. Anthrax vaccine immunization program (avip) update date signed. In addition to PA, aluminum hydroxide, small amounts. EF and LF, and other uncharacterized bacterial byproducts. The potency of vaccine lots is determined both by the. PA antibody titres by ELISA. Military troops have several problems and concerns with the vaccination program linked to mistrust. Anthrax bacteria infect people and animals when spores are inhaled, ingested. Surveillance Tools, Reserve Health Readiness Program, and more; Topics Submenu. Military Medical History National Museum of Health and Medicine. ANTHRAX VACCINATION IMMUNIZATION PROGRAM Proven Protection Against a Documented Threat STATEMENT BY Honorable RUDY deLEON Deputy Secretary of Defense Honorable David Oliver Principle Deputy Under Secretary of Defense. The US vaccine was used only by several thousand people until. There is also no data. Furthermore, the. UK and MDPH- PA vaccines is found to vary. 
Therefore, attempts have been ongoing. It. has been proposed that better vaccines would generate cell- mediated. Although virulence factors other than the toxin proteins and. These include a type 1. DNA topoisomerase on p. X0. 1 (2. 4), and chromosomally- encoded. Vaccine development. In the US, two general approaches toward an improved vaccine. First, a chemically pure. PA vaccine has been sought (4. One candidate has been derived from a. LF, EF and. spore, achieving 9. PA's biologic activity. Whether it will stimulate adequate immunity. The second approach seeks a live vaccine that. PA but also contains other immunogenic epitopes. Although a variety of. Part III of this series will examine the anthrax vaccines'. BW. REFERENCESAnaisimova TI, Pimenov TV, Kozhukhov VV et. Development of method for preparation and maintenance of. STI- 1 and test strain Zenkovsky. In Salisbury. Medical Bulletin, special supplement #8. June 1. 99. 6 p 1. Annas G: Changing the consent rules for Desert. Storm. NEJM 3. 26: 7. Anthony BF and Sutton A. The role of the. FDA in vaccine testing and licensure. In Lelvine MM, Woodrow. GC, Kaper JB et al. New. York, Marcel Dekker, Inc., 1. Anthrax vaccine is refused. Bull At Sci 4. 3: 4. Bernstein BJ: The birth of the US biological. Sci Am 2. 56: 1. 16, 1. Brachman PS and Friedlander AM: Anthrax. Philadelphia. WB Saunders, 1. Brachman PS, Gold H, Plotkin SA et al: Field. Am J Public Health 5. Broad W: Gene- engineered anthrax: is it a. New York Times, Feb 1. Broad W: Norway's 1. New York Times, June 2. A1. 1. Broster MG and Hibbs SE: Protective efficacy. Salisbury Medical. Bulletin, special supplement #6. January 1. 99. 0 p 9. Burrow GN: Letter to Rudy de Leon, Undersecretary for. Defense. 2/1. 9/9. Bussiere JL, Mc. Cormick GC and Green JD. Preclinical safety assessment considerations. Pharm Biotechnol. Butler D: Admission on Gulf War vaccines. Nature 1. 99. 7; 3. Committee on Veterans' Affairs, staff report. US Senate: Is military research hazardous to veterans' health? December 8. Committee on Government Reform and Oversight. Gulf War veterans' illnesses: VA, DOD continue to resist strong. US government printing office. Coulson NM, Fulop M and Titball RW: Bacillus. Salmonella typhimurium. SL 3. 26. 1, affords protection against anthrax spore challenge. Salisbury Medical Bulletin, special supplement. January 1. 99. 0 p 1. Duesbury NS, Webb CP, Leppla,SH et al: Proteolytic. MAP- kinase- kinase by anthrax lethal factor. Science. 2. 80: 7. Ezzell JW and Abshire TG: Immunological. Bacillus anthracis. Infect. Immun 5. 6: 3. Farchaus JW, Ribot WJ, Jendrek S et al. Fermentation, purification, and characterization of protective. Bacillus anthracis. Ann Int. Med 1. 21: 3. Food and Drug Administration. Report of. the Inspection of Michigan Biologic Products Institute (Form. FDA 4. 83), February 2. And other inspection reports from. Fouet A, Sirard J- C, Mock M: Virulence gene. Salisbury Medical Bulletin, special supplement. June 1. 99. 6 , p. Fox J. May, 9. 8 P. Friedlander AM, Welkos SL, Pitt MLM et al. Postexposure prophylaxis against experimental inhalation anthrax. Stockholm International Peace Research Institute, Oxford. University Press, 1. Princeton University Press, Princeton NJ, 1. Sailors refuse vaccine. Navy. Times, April 2. Z. Gold H: Studies on anthrax. J Lab Clin Med. p. Hambleton P and Turnbull PCB: Anthrax vaccine. Adv Biotechnol Processes 1. Heeren, RH: Anthrax in Louisiana. New Orleans. Medical and Surgical Journal 9. Holmes RK: Anthrax. In Fauci AS, Braunwald. E, Isselbacher KJ et al. New York, Mc. Graw- Hill, 1. Huxsoll DL, Patrick WC, Parrott CD: Veterinary. JAVMA 1. 90: 7. 14, 1. Ivins BE: Anthrax vaccines - - how stable. ASM 9. 8th General Meeting, May 1. Atlanta. Ivins BE, Ezzell JW, Jemski J et al: Immunization. Bacillus anthracis. Infect. Immun 5. 2: 4. Ivins BE, Fellows PF and Nelson GO: Efficacy. Bacillus anthracis. Vaccine. 12: 8. 72, 1. Ivins BE, Fellows PF, Pitt MLM: Efficacy. Bacillus anthracis. Salisbury Medical. Bulletin, special supplement no. Vaccine 1. 3: 1. 77. Ivins BE and Welkos SL: Recent advances. Eur. J Epidem 4: 1. Ivins BE, Welkos SL, Knudson GB et al: Immunization. Aro- ) mutants. of Bacillus anthracis and with recombinant strains of Bacillus. Infect Immun. 5. 8: 3. Ivins BE, Welkos SL, Little SF et al: Cloned. Salisbury Medical Bulletin, special supplement #6. January 1. 99. 0 p 8. Ivins BE, Welkos SL, Little SF et al: Immunization. Bacillus anthracis protective antigen combined. Infect Immun 6. 0: 6. Jackson P, Hugh- Jones ME, Adair DM et al. PCR analysis of tissue samples from the 1. Sverdlovsk anthrax. Bacillus anthracis strains. Proc Nat Acad Sci 9. Joint Vaccine Acquisition Program: Final. Programmatic Environmental Assessment. Joint Vaccine Acquisition. Program Project Management Office, Department of the Army, August. Jones MN, Beedham RJ, Turnbull PCB et al. Efficacy of the UK human anthrax vaccine in guinea pigs against. Bacillus anthracis. Salisbury Medical Bulletin. June 1. 99. 6 p 1. Jones MN, Beedham RJ, Turnbull PCB et al. Antibiotic prophylaxis for inhalation anthrax. Salisbury Medical. Bulletin, special supplement #8. June 1. 99. 6 p 1. Kadlec RP, Zelicoff AP and Vrtis AM: Biological. JAMA. 2. 78: 3. 51, 1. Kaufman A: Anthrax Vaccine Safety and Efficacy. Pro. MED- Mail. 4/1. Mil Med 1. 51: 7. Leppla SH, Friedlander AF, Singh Y et al. A model for anthrax toxic action at the cellular level. Salisbury. Medical Bulletin, special supplement #6. January 1. 99. 0 p 4. Leppla SH, Klimpel KR, Singh Y et al: Interaction. Salisbury Medical Bulletin. June 1. 99. 6, p. Lesnyak OT, Saltykov RA: Comparative assessment. Zh Mikrobiol Epidemiol Immunobiol. Lincoln RE, Walker JS, Klein F et al: Value. Fed Proc 2. 6: 1. Little SF, Ivins BE, Fellows PF et al: Passive. Bacillus anthracis. Infect Immun 6. 5: 5. Little SF and Knudsen GB: Comparative efficacy. Bacillus anthracis live spore vaccine and protective antigen. Infection and Immunity. PA News (UK) 9/2. Miller J: Clinton seeks additional $3. New York Times, June 9, 1. New York Times, July 8, 1. Physicians for Social Responsibility. Quarterly 2: 1. 98, 1. Nass M: Anthrax vaccine and the prevention. ASA Newsletter 1. Pro- MED Mail. May 7, 1. Pharm. Biotechnol 6: 1, 1. Norton- Taylor R: MOD ignored warning on. Gulf drugs. The Guardian (UK) October 2. Ora. Vax, Inc. Joins Dyn. Port In Department. Defense Contract To Develop FDA- Licensed Vaccines Against. Potential Biological Warfare Agents CAMBRIDGE, Mass., May 0. PRNewswire/ - - Ora. Vax, Inc. In Mandell GL, Bennett JE and Dolin R, eds. Principles. and practice of infectious diseases, 4th ed. Salisbury Medical. Bulletin, special supplement no. Salisbury Medical. Bulletin, special supplement #8. June 1. 99. 6, p 1. Pomerantsev AP et al: Expression of cereolysin. AB genes in Bacillus anthracis vaccine strain ensures protection. Vaccine 1. 5. 1. 84. Presidential Advisory Committee on Gulf. War Veterans' Illnesses. Interim Report, February, 1. Preston R: The Bioweaponeers. New Yorker. March 9, 1. Puziss M, Manning LC, Lynch JW et al: Large- scale. Bacillus anthracis in anaerobic. Appl Microbiol 1. Regan JC: The local and general serum treatment. JAMA 7. 7: 1. 94. Rodriguez PM. Anti- HIV mix is found in blood. Gulf War veterans. Washington Times, 8/2. Gulf War syndrome: is. Th. 2. profile? Lancet 1. Schlingman AS, Devlin HB, Wright GC: Immunizing. Bacillus. anthracis in cattle, sheep and swine. Am J Vet Res. 17: 2. Shlyakov E: Anthraxin- -a skin test for early. Salisbury Medical Bulletin, special supplement #8. June 1. 99. 6, p. Shlyakov EN and Rubinstein E: Human live. USSR. Vaccine 1. 2: 7. Shlyakov E, Rubinstein E, Novikov I: Anthrax. Salisbury Medical Bulletin. June 1. 99. 6, p. Stepanov AS and Leppla SH: Macrophages are. Bacillus anthracis inside and outside the host cell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2016
Categories |
 RSS Feed
RSS Feed
